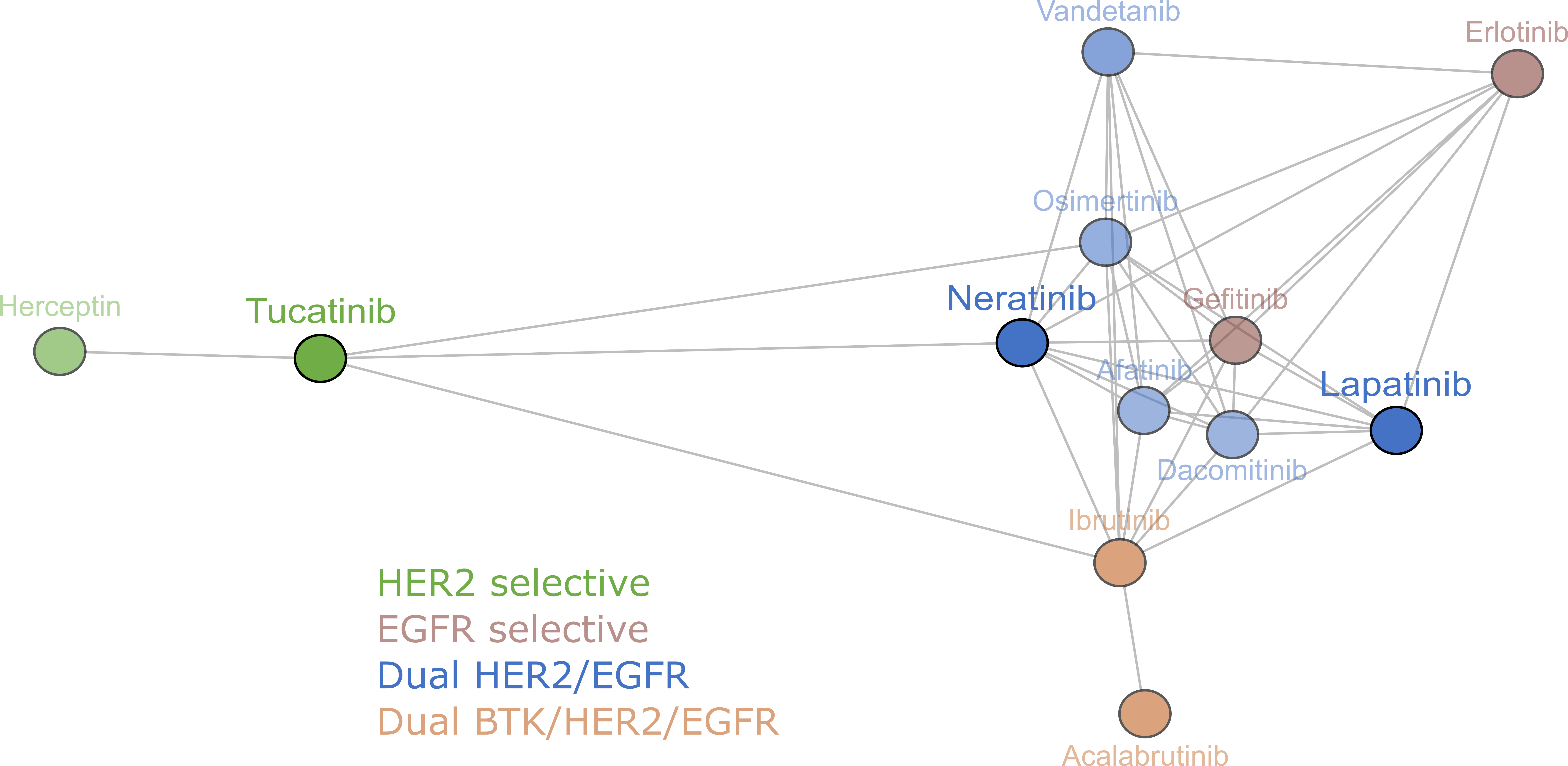
January 21, 2021: A new article on HER2-targeted tyrosine kinase inhibitors is now online in British Journal of Cancer (Conlon et al, 2021). The illustration shows clustering analysis of 12 EGFR and HER2 inhibitors on 115 cancer cell lines shows that the EGFR and HER2 inhibitors have similar cellular sensitivity profiles, confirming the selective nature of the drugs. Compounds (circles) are connected when the Pearson correlation is >0.5.
New in British Journal of Cancer: Comparative analysis of drug response and gene profiling of HER2-targeted tyrosine kinase inhibitors
Oss, Dublin, Los Angeles, January 21, – Three small molecule HER2 tyrosine kinase inhibitors have been approved for breast cancer treatment by the US Food and Drug Administration: lapatinib, neratinib and tucatinib. HER2-positive status is the only approved patient selection and clinical marker of response to these inhibitors. Novel biomarkers are required to define beneficial patient populations.
In a recent study published in British Journal of Cancer, Conlon et al. (2021) present a direct comparison of the anti-proliferative effects of the three inhibitors on a panel of 115 cancer cell lines. All three were effective against HER2-amplified breast cancer models; neratinib showing the most potent activity, followed by tucatinib then lapatinib. Neratinib displayed the greatest activity in HER2– and EGFR-mutant cells.
To determine the overall similarities and differences of the three HER2 tyrosine kinase inhibitors and other anti-cancer agents, Conlon et al. compared inhibitor IC50 fingerprints in the cancer cell line proliferation assays with those of 168 reference agents, including cytotoxic chemotherapeutics and many targeted inhibitors with diverse mechanisms of action (Uitdehaag et al., 2016). The IC50 profiles of the three HER2 tyrosine kinase inhibitors showed significant similarity with other tyrosine kinase inhibitors and not with cytotoxic drugs, confirming the selective nature of the therapies (see illustration on top).
The article by Conlon et al. also describes the identification of novel biomarkers of HER2 kinase inhibitor response. These were identified by correlating the anti-proliferative activity of the three HER2 inhibitors across the 115 cell line panel in an unbiased manner to mutations or copy number variations in well-known oncogenes and tumour-suppressor genes and basal expression levels of 18,900 genes. Neratinib was the most effective HER2-targeted tyrosine kinase inhibitor against ERBB2-amplified, -mutant, and EGFR-mutant cell lines. This analysis revealed novel resistance mechanisms that may be exploited using combinatorial strategies.

The paper in British Journal of Cancer is the result of a research collaboration between Dublin City University, Puma Biotechnology, Inc. and NTRC.
NTRC is a precision medicine company dedicated to discovering new anti cancer drug candidates. We help you to find a mechanistic hypothesis before entering the clinic. We can study a wide range of cancer cells, primary patient material and immune cells in vitro, in isolation and in coculture, after exposure to monotherapy and combination therapy. In addition, we perform in-depth mechanistic analyses in cells and by biophysical methods, such as Biacore and LC-MS/MS. Keywords are: Quality. Flexibility. Short Turnaround Time.





